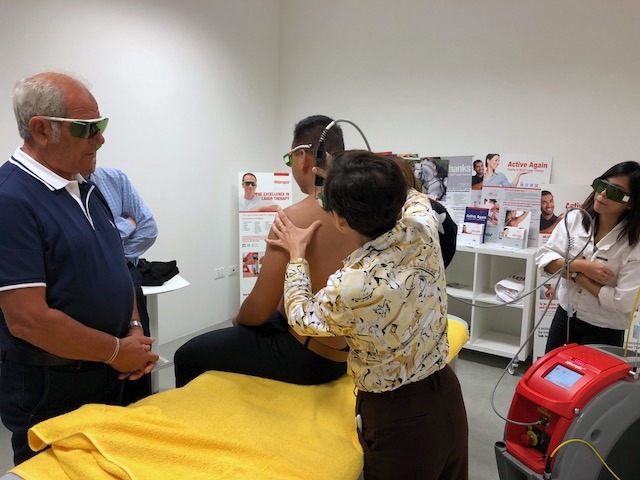
Full immersion training for Thai doctors in ASAlaser
A select group of Thai physicians specialised in physical therapy and rehabilitation from government facilities (Police and Thai Air Force hospitals) and from prestigious local universities attended a training session directly at the ASAlaser headquarters. The training, dedicated to examining in-depth the therapeutic solutions of the company (Hilterapia® and MLS® Laser Therapy), was divided into a theoretical and a practical section.
Three days of full immersion that enabled participants to learn the biological and therapeutic effects of the therapies and to thoroughly analyse the methods of application in order to optimise the obtainable results.
"It was this very aspect of improving the management of methods and schedules - explains Giacomo Granozio, Area Manager of ASAlaser – that particularly interested the doctors working in hospitals who, having to treat a large number of patients on a daily basis, needed concrete guidelines to be able to offer the best results whilst accelerating the schedules. The suggestions offered by our trainers for an advanced use of the therapies were counterchecked during the practical session. Some patients, including Enyob Faniel (gold-medal winner in the team marathon at the European Championships in Berlin), were treated in fact by applying these advanced methods which provide for a reduction in the time of application and energy administered".
The training for the Thai specialists does not stop here, however, confirming the importance that ASA attributes to continuous training: a new appointment, in fact, is scheduled during the sixteenth edition of Apalms (Asian Pacific Association for Laser Medicine and Surgery) from 19th to 20th October in Bangkok.