





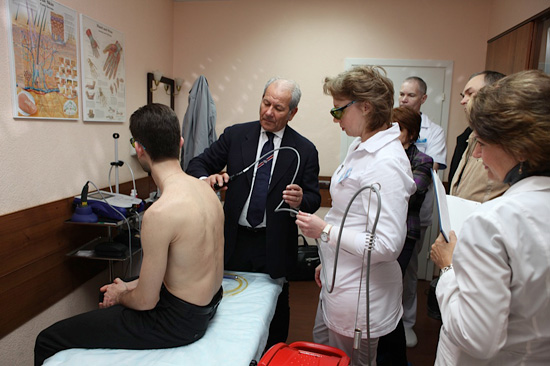
The hospital in Moscow "Dikul", specialized in orthopedics and rehabilitation, and the "Moscow University Municipal Hospital 31" were the venues of the continuous education centered on MLS® Laser Therapy and Hilterapia® kept by the staff of ASAlaser led by Lucio Zaghetto (CEO ASAlaser).
Participants, after having taken part in a course dedicated to Hilterapia® last December, are now confronting with the MLS® Laser Therapy, testing the concrete benefits.
"The 10 participants to "Master Class Hilterapia® and MLS®" - explains Lucio Zaghetto - after observing the absolute effectiveness of Hilterapia® compared to other therapeutic techniques, have now understood the importance of integrating it with the MLS®. In fact, if MLS® Laser Therapy is optimal for the resolution of diseases involving shallow areas, such as the neck pain, Hilterapia® is the most suitable solution to act in depth. ASAlaser objective is indeed to provide the most effective, safe and fast therapy to solve specific diseases".
The interest shown for the two treatments has kept pace with that for technical and scientific innovations that underlie them. "Following the "preliminary" course, viewing the different treatment protocols and related clinical research and seeing live results obtained with the first session conducted by their colleagues, the course participants showed amazement and curiosity up to wishing to investigate at once the application of the two therapies in the resolution of many illnesses. On the strength on these results, we will continue our program of continuous training, possibly in association with our courses online that aroused great appeal among people attending the "Master Class"".
Two of the most popular medical centers in Russia as well recognize the effectiveness of MLS® Laser Therapy and Hilterapia® confirming the success already collected in many Countries in the world including the United States. "A further confirmation of the interest for our therapies was obtained partecipating to the congress "Traumatology and orthopedics. Present and Future" in Moscow where our devices have attracted the attention of professionals".







L'accesso alla visualizzazione dei prodotti e al materiale informativo è riservato agli operatori del settore in ottemperanza alla legislazione vigente. ASA richiede di qualificarsi come operatore del settore per procedere con la navigazione.
Decreto Legislativo 24 febbraio 1997, n°46 Articolo 21
1. E' vietata la pubblicità verso il pubblico dei dispositivi che, secondo disposizioni adottate con decreto del Ministro della Sanità, possono essere venduti soltanto su prescrizione medica o essere impiegati eventualmente con l'assistenza di un medico o di altro professionista sanitario.
2. La pubblicità presso il pubblico dei dispositivi diversi da quelli di cui al comma 1 è soggetta ad autorizzazione del Ministero della Sanità. Sulle domande di autorizzazione esprime parere la Commissione di esperti prevista dall'articolo 6, comma 3, del decreto legislativo
30 dicembre 1992, n. 541, che a tal fine è integrata da un rappresentante del Dipartimento del Ministero della Sanità competente in materia di dispositivi medici e da uno del Ministero dell'Industria, del commercio e dell'artigianato.
Some of the contents of this website cannot be disclosed in the USA and its territories and possesions, for regulatory reasons. If you are a US resident, please click on the button here below and access ASA's distributor website for North America.